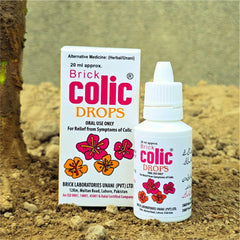
Brick Colic® Drops
Herbal Supplement
Short Description
A gentle herbal supplement traditionally used in Unani medicine to support digestive comfort and general wellness.
DRAP-Enlisted and crafted under certified standards.
Full Description
Brick Colic Drops is a DRAP-Enlisted herbal supplement formulated with botanicals traditionally used in Unani medicine to promote digestive wellness. It contains:
-
Group 1: Cinnamon, Cardamom, Fennel, Ginger – traditionally valued for their aromatic and carminative roles
-
Group 2: Hyoscyamus and Valerian – historically referenced for their calming properties in Unani formulations
This alcohol-free, plant-based formulation is manufactured under ISO 9001, ISO 14001, ISO 45001 & HALAL certified standards. It is suitable for general wellness use when taken as directed.
Key Benefits
- Traditionally used to support digestive wellness
- Contains botanicals studied for their aromatic and soothing properties
- May contribute to overall comfort and relaxation
- Plant-based formulation suitable for regular dietary use
Suggested Use
- Use under the supervision of a qualified healthcare professional
- Adults: dosage as advised by a practitioner
- For infants and children, dosage should be determined by a qualified practitioner
- Consistent use is recommended for best results
Packaging
20 ml dropper bottle
General Safety & Storage Instructions
- Keep out of reach of children
- Store below 30°C in a cool, dry place
- Protect from direct sunlight
- Shake well before use
- Use only under the supervision of a qualified healthcare professional
- If symptoms persist, consult a registered practitioner
- For general wellness use only; not a pharmaceutical medicine
Disclaimer
This product is a herbal supplement and not a pharmaceutical medicine.
It is marketed for general wellness and traditional use only.
It is not intended to diagnose, treat, cure, or prevent any disease.
Compliance & Regulatory Credentials
Manufacturer:
M/s Brick Laboratories Unani (Private) Ltd.
5-A, Ahbab Colony, 12 KM Multan Road, Lahore, Pakistan
Regulatory Enlistments:
- DRAP Manufacturer Enlistment #: 00461 (Form 6)
- DRAP Product Enlistment #: 00461.0001 (Form 7)
- GTIN (GS1): 8964002483006
- GS1 Company Prefix: 8964002483
Legal & Intellectual Property:
- National Tax Number (NTN): 1350562-9
- Trademark Registration #: NA
- Copyright #: NA
Certifications:
- ISO 9001 – Quality Management
- ISO 14001 – Environmental Management
- ISO 45001 – Occupational Health & Safety
- HALAL Certification
Memberships:
- The Lahore Chamber of Commerce & Industry (Membership No: 48881-A)
- Pakistan Tibbi Pharmaceutical Manufacturers Association (PTPMA) – Associate Class
Compliance Statement
This product is a HERBAL SUPPLEMENT under the HERBAL/UNANI CATEGORY, officially DRAP-Enlisted.
It is marketed for GENERAL WELLNESS and TRADITIONAL USE ONLY.
It is not a pharmaceutical medicine and is not intended to diagnose, treat, cure, or prevent any disease.
This listing complies with Google Merchant Center’s personalized advertising and healthcare policies.